A double replacement reaction takes place when two compounds exchange elements to form two new compounds.
The chemical formula they usually use is: AX + BY --> AY + BX
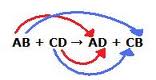
This is a picture of the exchange of two elements
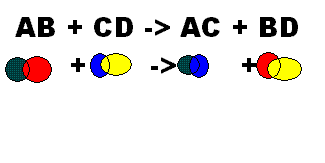
This example is showing that C and D replace each other.
Sources:
http://www.east-buc.k12.ia.us/03_04/CE1/KE/k_e5.htm
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Reactions_in_Aqueous_Solutions/Precipitation_Reactions
http://www.quia.com/jg/2145388list.html
http://www.east-buc.k12.ia.us/03_04/CE1/KE/k_e5.htm
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Reactions_in_Aqueous_Solutions/Precipitation_Reactions
http://www.quia.com/jg/2145388list.html
