A single replacement is when one element replaces another element in a compound.
A chemcial formula for this would be: A + BC --> BA + C (single anion replaces anion in compound) or
A + BC --> AC + B (single cation replaces cation in compound)
A + BC --> AC + B (single cation replaces cation in compound)
Sources:
http://sartep.com/chem/tutorials/tut.cfm?tutorial=Single+Replacement+Reactions&chap=6
http://thescienceclassroom.org/chemistry-lessons/chemical-quantities-and-reactions/types-of-chemical-reactions/
http://prezi.com/e9ez5cahv44p/4-types-of-chemical-reactions/
http://sartep.com/chem/tutorials/tut.cfm?tutorial=Single+Replacement+Reactions&chap=6
http://thescienceclassroom.org/chemistry-lessons/chemical-quantities-and-reactions/types-of-chemical-reactions/
http://prezi.com/e9ez5cahv44p/4-types-of-chemical-reactions/
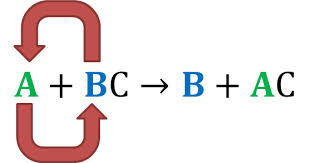
Here is an example of a single replacement reaction. In this picture A and B are being replaced with each other.
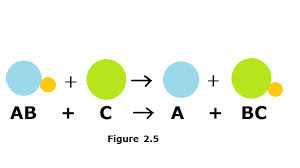
In this picture it shows the same thing happening!
Characteristic of a Single Replacement reaction: Single Replacement reactions are redox reactions.
